Acids and Bases
Name:_________________________________
Write the name for the two things you are comparing next to each of the circles below. Write words that describe how these two things are different in the areas that do not overlap. Write words that describe how these two things are similar in the area where the circles overlap.

1. Graphic Organizer #1
Chemists throughout the Years
Name:_________________________________
With the lists of Chemists given on the powerpoint, please lists the Chemists from left to right, left being the earlier dates to the right being closer to present date. Write the starting date on the left end and the ending date on the right side. If necessary, add other dates to the marks on the timeline. Sequence events in order on the timeline. Be sure to include a date and description for all events on the timeline.

2. Graphic Organizer #2
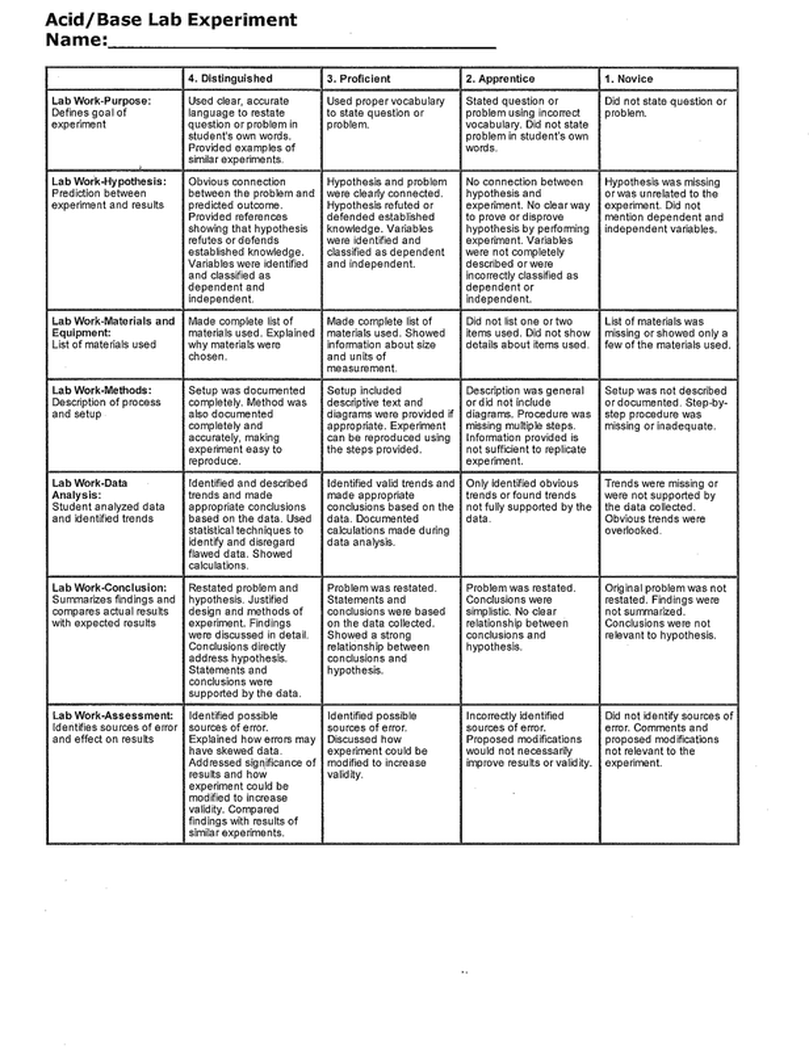
Acid/Base Lab Experiment
Name:_________________________________
In the Observation column, write about the thing you have observed and want to test. In the Hypothesis column, write a theory to explain the events you observed. In the Experiment column, describe the steps and materials you will use to test the hypothesis. In the Predictions column, write what you think will happen. In the Conclusion column, write a summary of what happened during the experiment and whether the hypothesis was true or false.
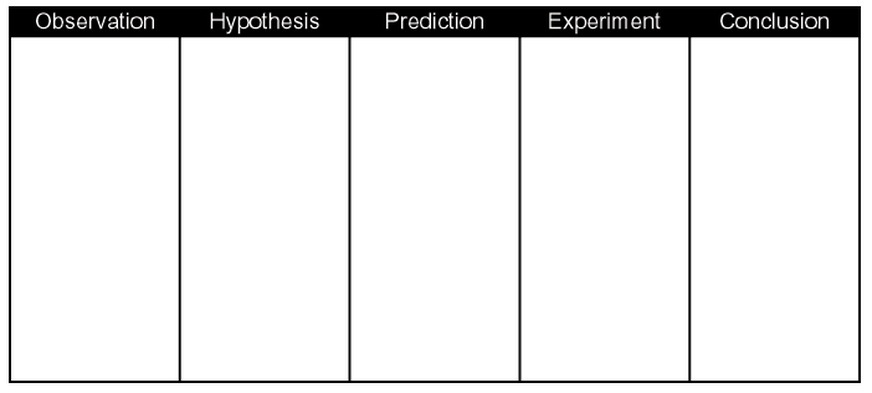
3. Graphic Organizer #3
Stoichiometry
Name:_________________________________
Think about all the steps in the process. Write the first step in the process in the First box. Write the next steps in their own boxes. Write the last step in the bottom box. If there are too many boxes on the page, skip one. If you need more boxes, number the steps and continue them on the back of the page.
Name:_________________________________
Think about all the steps in the process. Write the first step in the process in the First box. Write the next steps in their own boxes. Write the last step in the bottom box. If there are too many boxes on the page, skip one. If you need more boxes, number the steps and continue them on the back of the page.
Chemical Reactions
Name:_________________________________
Write about the word "Chemical Reactions," you are to describe in the center bubble. Write details about the word in the bubbles connected to it. Use the five senses and who, what, when, where, why, and how to think of more details.
Laboratory Safety Rules
Name:_________________________________
Think of the topic. In the K column, write the things that you already know about it. In the W column, write the things that you want to know about it. When you are done with research and experiments, write what you learned about it in the L column.
Name:_________________________________
Think of the topic. In the K column, write the things that you already know about it. In the W column, write the things that you want to know about it. When you are done with research and experiments, write what you learned about it in the L column.
Rules of Assigning Oxidation Numbers
Name:_________________________________
Write the main idea in the center bubble. Brainstorm related ideas in the connecting bubbles. Add details to the bubble connected to each idea. If you need more space, draw your own bubbles and connections.
Name:_________________________________
Write the main idea in the center bubble. Brainstorm related ideas in the connecting bubbles. Add details to the bubble connected to each idea. If you need more space, draw your own bubbles and connections.
Technology Lesson that was downloaded to help create the rubric for the lesson plan: http://www.madsci.org/experiments/archive/859332497.Ch.html
Cabbage Juice - pH indicatorArea of Science: Chemistry
Meant for at least Grade 4-6 (age 8-10).
This experiment is edible.
An adult need not be present.
Overview:
Make your own acid/base indicator by boiling red cabbage. Use the juice to pH different fluids.
Equipment:
1. 1/2 head of *red* cabbage or so.
2. Metal grater.
3. Pot filled with enough water to cover the grated cabbage.
4. Strainer
5. Some acid/base solutions: for an acid try vinegar, for a base - mix some detergent in water.
Safety:
Be careful with the fingers when grating the cabbage.
How to do the experiment:
1. Grate the cabbage into small pieces and place them in the pot + water.
2. Boil the mixture for 20-30 minutes, until the liquid turns a dark purplish color.
3. Decant the fluid into a glass or jar, pouring through a strainer to remove the cabbage. Save the cabbage.. mix with a little vinegar and you can eat it on hot dogs, etc.. The collected fluid should be bluish/dark purple in color.
4. Make up some 'test' acid/base solutions. A good acid to use would be white vinegar. You could also try soda water/sprite or diluted juice from a lemon or orange. You can make a basic solution by mixing some washing detergent in water, or by adding some baking soda (sodium bicarbonate) to water. It's useful to have a control solution of (neutral) water - distilled water is best if you have access to it.
5. Add a few drops of the cabbage juice to your solutions, and note any color changes. The juice should turn pink in acidic solutions, and green in basic solutions.
6. You can use the indicator on any other solutions of interest, or try drying it on coffee filters to create a simple form of "pH paper."
Explanation:
Red cabbage contains pigments call anthocyanins. The pigments give it the red/purplish color. Anthocyanins belong to group of chemical compounds called flavonoids.
For most pH indicators, the compound acquires a proton at low pH (lots of H+) but looses it at higher pH. This seemingly minor alteration is sufficient to alter the wavelengths of light reflected by the compound, thus creating the color change with respect to pH. Anthocyanins behave somewhat inversely in that the pigments "gain" an -OH at basic pH, but loose it at acidic pH. The WWW link below describes the chemistry with structures if you want to see the details.
The chemistry behind pH, acids and bases.. An acidic solution contains an excess of protons or H+. pH is a measure of how 'acidic' a solution is. The lower the pH, the more acidic the solution. In chemical terms, pH means "the negative log of the concentration of protons" in solution. Chemistry students should recognize this as pH = -log[H+]. If the concentration of H+ is .01M, the pH will be:
-log[.01] = -log[10^-2] = -(-2) = 2 (very acidic!).
"Neutral" solutions (water, e.g.) have a pH of 7. This number coicides with the amount of H+ naturally formed in water from the equilibrium reaction: H2O <--> H+ + OH- (H+ experimentally known to be ~10^-7M; OH- is also the same concentration). "Basic" solutions have a pH greater than 7 - meaning they have less free H+ than that of neutral water.
Useful References:
General anthocyanin structure
Anthocyanins and pH
Cabbage Juice - pH indicatorArea of Science: Chemistry
Meant for at least Grade 4-6 (age 8-10).
This experiment is edible.
An adult need not be present.
Overview:
Make your own acid/base indicator by boiling red cabbage. Use the juice to pH different fluids.
Equipment:
1. 1/2 head of *red* cabbage or so.
2. Metal grater.
3. Pot filled with enough water to cover the grated cabbage.
4. Strainer
5. Some acid/base solutions: for an acid try vinegar, for a base - mix some detergent in water.
Safety:
Be careful with the fingers when grating the cabbage.
How to do the experiment:
1. Grate the cabbage into small pieces and place them in the pot + water.
2. Boil the mixture for 20-30 minutes, until the liquid turns a dark purplish color.
3. Decant the fluid into a glass or jar, pouring through a strainer to remove the cabbage. Save the cabbage.. mix with a little vinegar and you can eat it on hot dogs, etc.. The collected fluid should be bluish/dark purple in color.
4. Make up some 'test' acid/base solutions. A good acid to use would be white vinegar. You could also try soda water/sprite or diluted juice from a lemon or orange. You can make a basic solution by mixing some washing detergent in water, or by adding some baking soda (sodium bicarbonate) to water. It's useful to have a control solution of (neutral) water - distilled water is best if you have access to it.
5. Add a few drops of the cabbage juice to your solutions, and note any color changes. The juice should turn pink in acidic solutions, and green in basic solutions.
6. You can use the indicator on any other solutions of interest, or try drying it on coffee filters to create a simple form of "pH paper."
Explanation:
Red cabbage contains pigments call anthocyanins. The pigments give it the red/purplish color. Anthocyanins belong to group of chemical compounds called flavonoids.
For most pH indicators, the compound acquires a proton at low pH (lots of H+) but looses it at higher pH. This seemingly minor alteration is sufficient to alter the wavelengths of light reflected by the compound, thus creating the color change with respect to pH. Anthocyanins behave somewhat inversely in that the pigments "gain" an -OH at basic pH, but loose it at acidic pH. The WWW link below describes the chemistry with structures if you want to see the details.
The chemistry behind pH, acids and bases.. An acidic solution contains an excess of protons or H+. pH is a measure of how 'acidic' a solution is. The lower the pH, the more acidic the solution. In chemical terms, pH means "the negative log of the concentration of protons" in solution. Chemistry students should recognize this as pH = -log[H+]. If the concentration of H+ is .01M, the pH will be:
-log[.01] = -log[10^-2] = -(-2) = 2 (very acidic!).
"Neutral" solutions (water, e.g.) have a pH of 7. This number coicides with the amount of H+ naturally formed in water from the equilibrium reaction: H2O <--> H+ + OH- (H+ experimentally known to be ~10^-7M; OH- is also the same concentration). "Basic" solutions have a pH greater than 7 - meaning they have less free H+ than that of neutral water.
Useful References:
General anthocyanin structure
Anthocyanins and pH